Research Facilities
Electrophysiology
We study the intrinsic and synaptic transmembrane ionic currents of neurons. Many recording techniques are used including various configurations of the patch-clamp technique usually in acute brain slices. We compare naïve and experimental pathological models in order to determine the ionic current(s) altered that are the cause of the disorder and a possible therapeutic target.
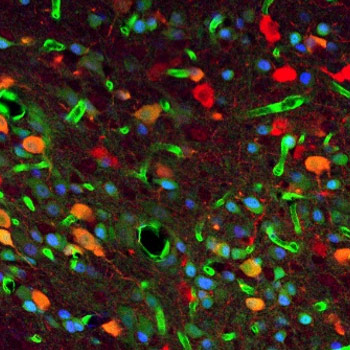
ImmunoHistoChemistry
Immunohistochemistry is a method of detecting a protein (or antigen) in sections of biological samples by means of antibodies directed specifically against this target protein. It is then possible to visualize it using markers coupled directly or indirectly to the antibodies. When using fluorescent dyes or fluorochromes as markers, the technique is called immunofluorescence. Under a microscope, and thanks to the fluorescence emission after excitation of the fluorochromes used, this technique makes it possible to reveal the presence or absence of a specific protein as well as its location in the cells of the tissue studied. There are different fluorochromes, each with a different excitation wavelength, allowing color combinations and therefore the detection of different proteins on the same control or pathological tissue.
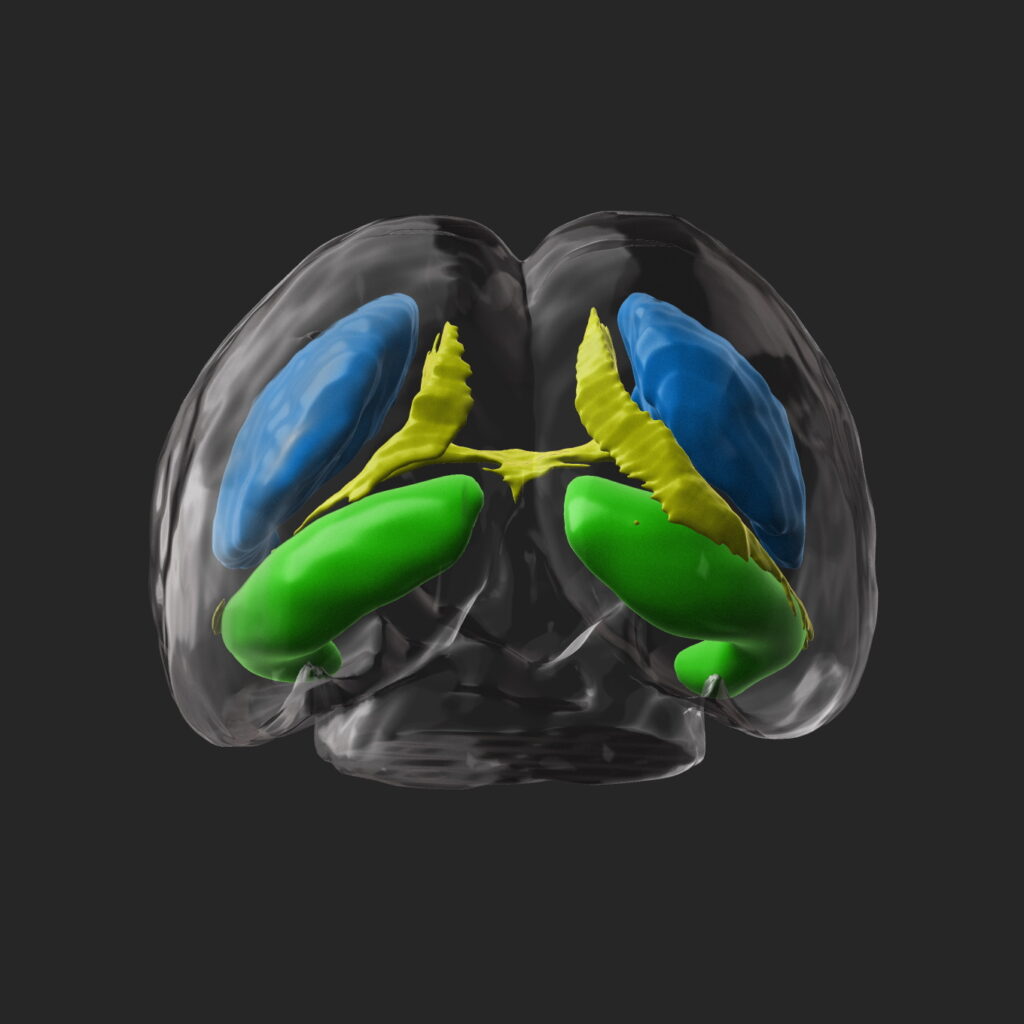
Clearing
The exploration of organs as a whole and at cellular resolution represents a challenge in biology due to the opacity properties of the tissues that make deep imaging difficult. The transparency technique enables to overcome this phenomenon by modifying the optical properties of light scattering and absorption at the tissue level, thus enabling volumetric imaging and facilitating the visualization of immunohistochemical markers. We use the iDisco transparency protocol to understand the establishment of major neuronal pathways and their alteration in a pathological context during embryonic and postnatal brain development in rodents.
The transparent brains are then analyzed using a light sheet microscope with the specific illumination of the sample by a sheet of light. We analyze a variable sequence of images in the acquisition plane, which can be assembled in stacks and then visualized in 3D using a specific software.
We have developed automated programs that enable to segment brain structures, measure their volume, count the number of cells, and evaluate their distribution. The results obtained in control animals serve us as a reference to highlight possible alterations in animal models reproducing neurodevelopmental pathologies.

Behavior
There are a wide variety of behavioral tests available for laboratory rodents, from tests of basic locomotor and sensory function to more complex behavior related to cognition, anxiety and emotion. The Behavior platform is equipped for a battery of tests adapted to the mouse models of neuropsychiatric disorders including Autism, and neurodegenerative disorders like Parkinson’s disease. These tests may be used as a package for phenotyping genetically modified mice or to screen for potential therapeutics.
Neurodevelopmental Tests:
These tests are used to characterize early markers of behavior in order to determine the neurobiological cause of these behaviors and thus assess the impact of an early prenatal or postnatal complication. Our routinely used tests include social interaction tests, ultrasonic vocalization test, stereotyped and repetitive behavior measurements, and motor system tests such as the cylinder test, the “open field” test, the “rotarod” or the “pole test”.
